NCERT Solutions for Science Class 9 Chapter 1
Chapter 1: Matters In Our Surroundings
Excercise 1:
Question 1: Which of the following are the matter?
Chair, air, love, smell, hate, almonds, thought, cold, cold drink, smell of perfume.
Answer: Matter is anything that occupies space and has some mass. There are three states of matter called Solid, Liquid and Gas. On the basis of these three states, we can define that which of this is a matter:
- Chair and almond are said to be in a solid state of matter as these have fixed shape.
- Cold drink is in liquid state as it has the tendency to flow.
- Air and the smell of perfume have gaseous particles which are free to move so this will also be considered as a gaseous state of matter.
- While Love, hate, cold, smell and thought are not having any mass or neither do they occupy space these are just emotions or sensations felt by human beings so they are not considered as matter.
Question 2: Give reasons for the following observation:
The smell of hot sizzling food reaches you several meters away, but to get the smell from cold food you have to go close.
Answer: The smell of hot sizzling food prepared by our mom reaches to us in our room from kitchen but if the food gets cold after some time, we did not feel any smell of that food this phenomenon can be defined on the basis of rate of diffusion which gets increases when the temperature get increases as high temperature increases the kinetic energy of food particles to get diffused in air. The temperature of hot food particles is high as compared to old one so its molecules get easily diffused in the air as compared to cold ones.
Question 3: A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Answer: This can be explained on the properties of matter to attract the particles towards themselves and this will also decide their shape and rigidity. The force of attraction is highest in case of solid as compared to liquid and gas this defines that particles of solid are tightly bound to each other. While in case of liquid particles they have less forces of attraction which defines that there is space between the particles and due to this reason that we can cut them easily. That is why we can say that due to less forces of attraction between water molecules a diver is able to cut through water in a swimming pool.
Question 4: What are the characteristics of particles of matter?
Answer: Matter is anything that occupies space and has some mass. There are three states of matter called Solid, Liquid and Gas.
The main characteristics of matter can be described as follows:
- Particles of matter have space between them and the order of spacing is highest in gas after that liquid and solid have very less space between their particles.
- Particles of matter are continuously moving in all the three states of matter.
- Particles of matter attract each other with strong forces which help them to bind with each other. In solid particles attraction is very high whereas in liquid it is low and in gases it is quite low.
Exercise 2:
Question : The mass per unit volume of a substance is called density (density = mass/volume). Arrange the following in order of increasing density − air, exhaust from chimney, honey, water, chalk, cotton, and iron.
Air < Exhaust from chimney < Cotton < Water < Honey < Chalk < Iron.
A. Tabulate the differences in the characteristics of states of matter.
Answer: Matter is anything that occupies space and has some mass. There are three states of matter called Solid, Liquid and Gas.
| Property | Solid State | Liquid State | Gaseous State |
| Shape and volume | It has a fixed shape and volume. | It has no definite shape unless it takes the shape of the vessel in which they are kept. It has a fixed volume. | It does not contain any shape or not have any fixed volume. |
| Compressibility | It is incompressible in nature. | It is compressible but to a smaller extent. | It is highly compressible in nature. |
| Forces of attraction | The force of attraction is highest in case of solids. | The force of attraction between liquid particles is less than solid particles but greater than gas. | The force of attraction in this case is very low. |
| Randomness | Particles of solid have no tendency to move freely therefore, these are not termed to be random. | Liquid particles have a tendency to move freely therefore these are random in nature. | Gaseous particles are very free to move so these are also random in nature. |
B. Comment upon the following: rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy, and density.
Answer: Rigidity: It is defined as the tendency of matter to resist a change in shape.
Compressibility: The ability of matter to reduce in volume when any type of external force is applied on it.
Fluidity: Tendency of particles to flow this property can be seen in case of liquid and gases which can also be known as fluids.
Filling a Gas Container: Gases neither have definite shape nor have definite volume. Gases take the shape of that container in which it gets filled. Hence by filling a gas container, it means the attainment of shape of the container by the gas.
Shape: Shape corresponds to fixed volume and boundary. Only solids have a fixed shape.
Kinetic Energy: Particles which produce energy possessed due to its continuous motion.
Density: It is defined as the mass per unit volume.
Question 3: Give Reasons:
a. A Gas Fills Compeletely the Vessel in Which it is Kept.
Answer: The gas particles have a tendency to move freely in all directions as they have very less force of attraction between their particles. Like water, gas can also take the shape of the container in which it is kept. Therefore, we can say that gas completely fills the vessel in which it is kept.
b. A Gas Exerts Pressure on the Walls of the Container.
Answer: The gas particles move freely due to its lesser forces of attraction between the particles. Therefore, these gaseous particles continuously collide with each other and with the walls of the container with a greater force. Pressure is known as the force produced by the gas particles per unit area. By this we can say that gas exerts pressure on the walls of the container.
c. A wooden table should be called a solid.
Answer: A wooden table is very rigid in nature which means that it has a definite shape and its shape cannot be changed easily and has definite volume too. The shape is fixed due to strong intermolecular forces hence it attains all the properties of solid therefore, it is considered as a solid.
d. We can easily move our hands in air, but to do the same through a solid block of wood, we need a karate expert.
Answer: Air particles have very less forces of attraction between their particles so they have large space in between them. But wood has very little space between the particles due to its high force of attraction therefore, wood is considered to be of rigid nature. Due to this reason, we can easily move our hands in air but the same will not happen through a solid block of wood. For this we need a karate expert.
Question 4: Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why.
Answer: As we know that density is defined as the mass per unit volume. This corresponds with the increase of the volume of any substance density will decrease as they are inversely proportional to each other.
Ice is solid in nature therefore it contains strong intermolecular forces which tightly bound them and they contain lesser volume but on the other hand liquid has tendency to move freely due to weak intermolecular forces and contain large volume.
From this we can say that water has larger volume and lesser density so it has a tendency to float on water.
Exercise – 3
Question 1: Convert the following temperature to Celsius scale:
A. 300 K
Answer: Celsius and Kelvin are two main scales to measure the temperature. By subtracting the 273K from the given value we can get the value in degrees Celsius. The formula corresponds to degree Celsius can be shown as:
\(X^0\space C(X-273)\)
\(300\space K=(300-273)^0\space C=27^\space C\)
B. 573 K
Answer: \(773K=(573-273)^0\space C=300^0\space\)
Question 2: What is the physical state of water at:
A. \(250^0\space C\)
Answer: Physical state corresponds to the state of matter whether it exists in solid, liquid or gas.
\(250^\space C\)– As we know that water starts boiling at \(100^0\space C\) and above this temperature water
exists in gaseous state. So, this defines that water at \(250^0\space C\) exists in a gaseous state.
B. \(100^0\space C\)
Answer: \(100^0\space C\) – It is the starting point where water starts boiling so at this temperature water exists in both liquid and gaseous states.
Question 3: For any substance, why does the temperature remain constant during the change of state?
Answer: This can be explained as the whole heat which we are providing to the substance to increase its temperature is used to break the intermolecular forces of attraction between them. This heat will also correspond to the latent heat i.e., the heat which gets absorbed or released during change of state. Hence all the energy gets used so temperature remains constant.
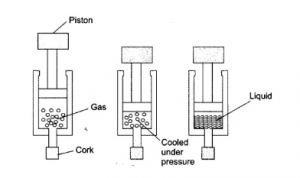
Question 4: Suggest a method to liquefy atmospheric gases.
Answer: Atmospheric gases can be defined as the gases present in the atmosphere. It can be liquified i.e., converted into liquid by applying suitable conditions of applying pressure and by reducing their temperature.
Exercise -4
Question 1: Why does a desert cooler cool better on a hot dry day?
Answer: This can be explained by the process called evaporation, this is the process in which the liquid particles absorb energy from the surroundings and cause cooling. The rate of evaporation generally depends on the amount of water vapour present in the air. If the amount of water vapor present in air is more than the rate of evaporation is more or vice-versa. On a hot dry day, the amount of water vapor present in air is quite low so this will evaporate easily and make its surroundings cooler. Thus, from this we can say that a desert cooler cools better on a hot dry day as compared to rainy one.
Question 2: How does water kept in an earthen pot (matka) become cool during summers?
Answer: An earthen pot or matka is generally made up of sand particles in which many tiny pores exist and this helps the water inside the pot to evaporate and surroundings makes the water cool. This is the reason why people kept the water in an earthen pot during summers.
Question 3: Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Answer: Acetone, petrol or perfume are considered as organic compounds which are volatile in nature whereas volatile substances are those which easily get vaporized and go through the process of evaporation. We know that during the process of evaporation particles of these organic liquids absorb energy from the surroundings or the surface of the palm and make the surroundings or surface of the palm somewhat cool. This is the reason why our palm feels cold when we put some acetone, petrol or perfume on it.
Answer: Why are we able to sip hot tea or milk faster from a saucer than a cup?
Answer: This can also be explained on the basis of rate of evaporation as we know that evaporation produces a cooling effect and evaporation depends on the surface area, larger the surface area higher the evaporation. As in saucer the area is larger as compared to cup so evaporation will be high in case of greater surface area. Thus, we can say that liquid cools faster in a saucer than in a cup and due to this reason, we are able to sip hot tea or milk faster from a saucer than a cup.
Question 5: What type of clothes should we wear in the summer?
Answer: In summer we usually sweat so we have to wear cotton or light-colored clothes because cotton or light-colored clothes can absorb more sweat from our body and transfers the sweat which is in the form of liquid to the atmosphere and makes the evaporation process faster. Evaporation process causes a cooling effect which makes our body cool in cotton clothes as compared to synthetic or woolen ones.
Question 1: Convert the following teperature to Celsius scale:
A. 300 K
Answer: Celsius and Kelvin are two main scales to measure the temperature. By subtracting the 273K from the given value we can get the value in degrees Celsius. The formula corresponds to degree Celsius can be shown as:
\(X^0\space C=(X-273)\)
\(300\space K=(300-273)^0\space C=27^0\space C\)
B. 573 K
Answer: \(573K=(573-273)^0\space C=300^0\space C\)
Question 2: Convert the following teperature to Kelvin scale:
A. \(25^0\space C\)
Answer: Celsius and Kelvin are two main scales to measure the temperature. By adding the 273K from the given value we can get the value in degrees Celsius. The formula corresponds
to degree Celsius can be shown as:
\(060\space C=273\space K\)
\(27^0\space C=(27+273)K=300\space K\)
B. \(373^0\space C\)
Answer:
\(373^0\space C=(373+273)\space K=646\space K\)
Question 3: Give reasor for the following obsevations.
A. Naphthalene balls disappear with time without leaving any solid.
Answer: This phenomenon can be explained on the basis of sublimation which defines that solid is directly converted into gaseous form without turning it into liquid. Naphthalene is
one that substances which undergo through the process of sublimation easily at room temperature. That is why we can say that naphthalene balls disappear after some time without leaving any solid.
B. We can get the smell of perfume sitting several meters away.
Answer: Gaseous particles have very less internuclear forces due to which its molecules are very free to move and it possesses high kinetic energy. Due to this reason particles of
perfumes diffuse into the atmosphere and its molecules will mix in the environment which enables us to smell the perfume from several meters away.
Question 4: Arrange the following substances in increasing order of forces of attraction between particles − water, sugar, oxygen.
Answer: Forces of attraction is the attracting power of molecules which keep them together and intermolecular forces are very strong in case of solid as compared to liquid or gas.
Here Sugar is said to be solid and contains higher forces of attraction.
Water is liquid comparatively containing lesser forces of attraction but higher as compared to gases.
Oxygen is a gas which contains very less attraction between forces.
Thus, the increasing order of forces of attraction between the particles of water, sugar and oxygen is Oxygen < Water < Sugar
Question 5: What is the physical state of water at:
\(25^0\space C\)
Answer: Physical state corresponds tot the state of matter whether it exists in solid, liquid or gas.
\(25^0\space C\) – As we know that ater melts at \(0^0\space C\) and abobe this teperature water exusts in liquid state. So, this defines that water at \(25^0\space C\) exists in a liquid state.
B. \(0^0\space C\)
Answer: \(0^0\space C\)- It is the temperature at which water starts melting i.e., water gets converted into liquid from ice so at this temperature water exists as both solid and liquid state.
C. \(100^0\space C\)
Answer: It is the starting point where water starts boiling so at this temperature water exists in both liquid and gaseous states.
Question 6: Give two reasons to justify:
A. Water at room temperature is a liquid.
Answer: We find water is in liquid state at room temperature this can be justified as follows:
- Water does not have any fixed shape; it can take the shape of the container in which it is kept and water has definite volume.
- It has a tendency to flow.
- Have weak intermolecular forces between their particles.
These all describe the property of liquid so we can say that water is liquid at room temperature.
B. An Iron Almirah is a Solid at Room Temperature.
Iron almirah kept in our room is said to be solid due to following reasons:
1. It has a fixed shape and definite volume.
2. It contains strong intermolecular forces between their particles.
3. Rigid in nature, difficult to compress.
These all describe the property of solid so we can say that almirah is solid at room temperature.
Question 7: Why is ice at 273 K more effective in cooling than water at the same temperature?
Answer: Here condition given that both ice and water are at same temperature i.e. 273 K. But ice at 273 K has less energy as compared to water this can be explained on the basis of latent heat of fusion which is possessed by water as an additional energy but ice does not have such type of energy. Therefore, we can say that at 273 K ice is more effective in cooling as compared to water.
Question 8: What produces more severe burns, boiling water or steam?
Answer: Steam and water both are said to be at the same temperature i.e., 373 K. But steam contains more energy as compared to boiling water. This can be explained on the basis of
latent heat of fusion which is possessed by water as an additional energy. Therefore, steam produces more severe burns than boiling water.
Question 9: Name A, B, C, D, E and F in the following diagram showing change in its state.
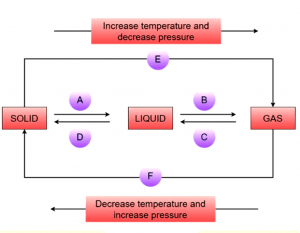
Answer:
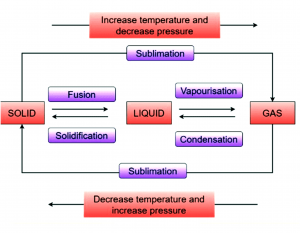
A is the process of converting solid into liquid is called Melting.
B is the process which converts liquid into gaseous state, this is called vaporisation.
C in which gases get converted into liquid this is called condensation.
D is the process which converts liquid into solid. It is called solidification.
E and F are the processes which convert solid into gas or vice versa is known as sublimation
Study materials
- Refernce Books
- NCERT Solutions
- Syllabus










