NCERT Solutions for Science Class 10 Chapter 4
NCERT Solutions for class-10 Science chapter 4 “Carbon and its compounds” is curated by senior and experienced teachers of EduAid. Before answering the questions of Chapter 4 , read the theory of the chapter multiple times and get a clear understanding of the concepts included in the chapter.
Here are the answers of the questions included in the NCERT book. Before diving deeep into the answers, try to attempt the questions by yourself.
Answer The Following Questions
1. What would be the electron dot structure of carbon dioxide which has the formula of \(CO_2\)?
Solution :
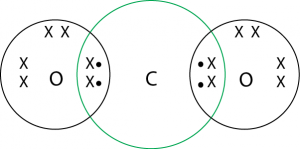
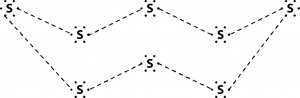
2. What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur?
Solution :
3. How many structural isomers can you draw for pentane?
Solution : The isomers are as under:
(i) ![]()
(ii) ![]()
(iii) 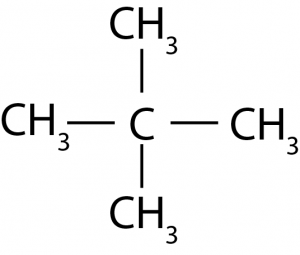
4. What are two properties of carbon which lead to the huge number of carbon compounds we see around us?
Solution : The two properties are :
(a) catenation- the ability to form bonds with other atoms of carbon.
(b) Tetravalancy of carbon.
5. What will be the formula and electron dot structure for cyclopentane?
Solution : Formula of cyclopentane is \(C_5H_{10}\). The electron dot structure cyclcopente is:
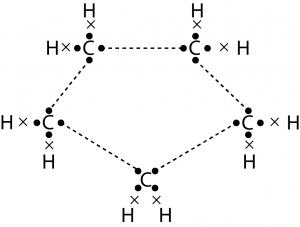
6. Draw the structure for the following compounds :
(i) Ethanoic acid
(ii) Bromopentane
(iii) Butane
(iv) Hexanal
Solution : Structure of compounds are given as under :
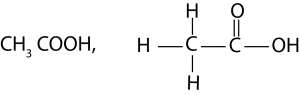
(i) Ethanoic acid
(ii) Bromopentane
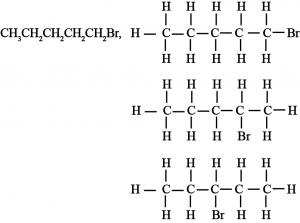
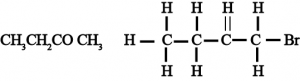
(iii) Butanone
(iv) Hexanal
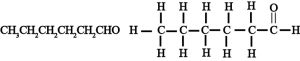
7. How would you name the following compounds?
(i) ![]()
(ii) ![]()
(iii) 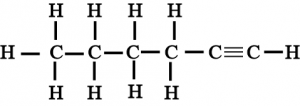
Solution :
(i) Bromomethane
(ii) Methanal (Formaldehyde)
(iii) Hexyne
8. Would you be able to check if water is hard by using a detergent?
Solution : No, we would be able to check if water is hard by using a detergent.
9. People use a variety of methods to wash clothes. Usually after adding the soap, they beat the clothes on stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes?
Solution : Agitation is necessary to obtain complete mecells formation and the emulsion of oil in water so that the whole of dirt is removed on rinsing with water.
10. Ethane, with the molecular formula \(C_2H_6\) has
(a) 6 covalent bonds
(b) 7 covalent bonds
(c) 8 covalent bonds
(d) 9 covalent bonds
Solution : (b) 7 covalent bonds
11. Butanone is a four carbon compound with the functional group
(a) carboxylic acid
(b) aldehyde
(c) ketone
(d) alcohol
Solution : (c) Ketone
12. While cooking, if the bottom of the vessels is getting blackened on the outside, it means that
(a) the fuel is not cooked completely.
(b) the fuel is not burning completely.
(c) the fuel is wet.
(d) the is burning completely.
Solution : (b) the fuel is not burning completely.
13. Explain the nature of the covalent bond using the bond formation in \(CH_3Cl\).
Solution : Covalent bond is formed by sharing of electrons between two atoms. It is non-ionic in nature.
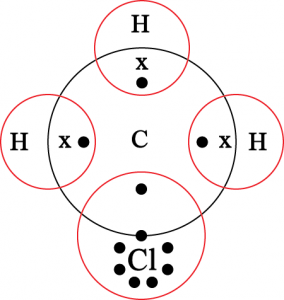
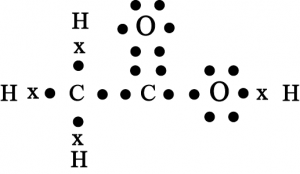
14. Draw the electron dot structure for
(a) \(Ethanoic\space acid\)
(b) \(H_2S\)
(c) \(Propanone\)
(d) \(F_2\)
Solution :
(a) Ethanoic acid
(b) \(H_2S\)
![]()
(c) Propanone
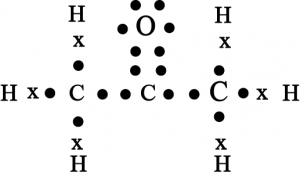
(d) \(F_2\)
![]()
15. What is a homologous series? Explain with an example.
Solution : Series of compounds in which the same functional group substitutes for hydrogen in a carbon chain is called homologues series. The difference between the formulae of any two successive members is \(–CH_2\) and difference between the molecular formula is \(14\space u\).
16. How can ethanol and Ethanoic acid be differentiated on the basis of their physical and chemical properties?
Solution : On the basis of physical properties :
Melting and boiling points of ethanol is \(156\space K\) and \(351\space K\) but melting and boiling point of Ethanoic acid is \(290\space K\) and \(391\space K\) respectively.
On the chemical properties :
Ethanoic acid reacts with sodium hydrogen carbonate liberating carbon dioxide while ethanol does not.
17. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Solution : Soap is sodium or potassium salt of long chain fatty acid. Two ends of soap molecules have different properties. The ionic end is hydrophilic. It dissolve in water while the hydrogen chain is hydrophobic, it dissolve in hydrocarbon. The hydrocarbon chains are oriented towards the oil droplet while the ionic ends are oriented towards water. Micelles formation will not take place in ethanol.
18. Why are carbon and its compounds used as fuels for most applications?
Solution : Carbon on combustion gives carbon dioxide and water. This reaction is accompanied by evolution of heat and light. The same is true for compounds of carbon. That is why carbon and its compounds are used as fuel for most applications.
19. Explain the formation of scum when hard water is treated with soap.
Solution : Hard water contains hydrogen carbonates, chlorides and sulphates of calcium and magnesium which reacts with soap to form scum. For example, calcium chloride reacts with soap to form scum.
Sodium stearate + Calcium chloride à sodium chloride + Calcium stearate(scum)
20. What change will you observe if you test soap with litmus paper (red and blue)?
Solution : Soap is sodium or potassium salt of fatty acid. It is obtained by treating of oil with caustic soda. Sodium stearate is thus a salt of weak acid and strong base. Its water solution will be slightly alkaline and will turn red litmus red.
21. What is hydrogenation? What is its industrial application?
Solution : Unsaturated hydrocarbons add hydrogen in presence of catalysts such as palladium or nickel to give saturated hydrocarbons. This process is called hydrogenation.
It is commercially used for converting vegetable oils to ‘vanaspati’ ghee in presence of nickel as catalyst.
22. Which of the following hydrocarbons undergo addition reactions?
\(C_6,\space C_3H_8, \space C_3H_6,\space C_2\) and \(CH_4\)
Solution : \(C_3H_6\) and \(C_2H_2\) will undergo addition reactions.
23. Give a test that can be used to differentiate chemically between butter and cooking oil?
Solution : Butter and cooking oil can be differentiated with the help of bromine water test. Cooking oil will decolorize the red colour of bromine water on shaking while butter will not.
24. Explain in mechanism of the cleaning action of soap.
Solution : Soap are sodium or potassium salt of fatty acids. Two ends of molecules of soap behave differently. This ionic end is hydrophilic and it is oriented towards water. The other hydrocarbon end is hydrophobic and it is oriented towards dirt which is oily in nature. A micelle formation around the oily dirt takes place. When flushed with excess of water, the micelle containing the dirt is removed, thus cleaning the clothes, etc.
25. Why do ionic compounds have high melting points?
Solution:
Ionic compounds have strong electrostatic forces of attraction between the ions. Therefore, it requires a lot of energy to overcome these forces. That is why ionic compounds have high melting points.
26. What are oxidising agents?
Solution:
Some substances such as alkaline potassium permanganate and acidified potassium dichromate are capable of adding oxygen to others. These are known as oxidising agents.
27. In the electrolytic refining of a metal M, what would you take as the anode, the cathode and the electrolyte?
Solution:
In the electrolytic refining of a metal M:
Anode → Impure metal M
Cathode → Thin strip of pure metal M
Electrolyte → Solution of salt of the metal M
Study materials
- Refernce Books
- NCERT Solutions
- Syllabus
Send Us A Message
Latest posts









